Last Updated: 26 Aug 2025
Source: Statifacts
By clicking “Accept All Cookies” you agree to the storing of cookies on your device to enhance site navigation, analyze site usage, and assist in our marketing efforts.
Privacy Policy
In Vitro Diagnostic (IVD) Reagents Market (By Type: Immune Diagnosis, Clinical and Biochemical, Molecular Diagnosis, POCT, and Other; By Application: Hospital, Laboratory, and Other; By Region: North America, Europe, Asia Pacific, Latin America, and Middle East & Africa) Industry Size, Share, Growth, Trends 2025 to 2034.
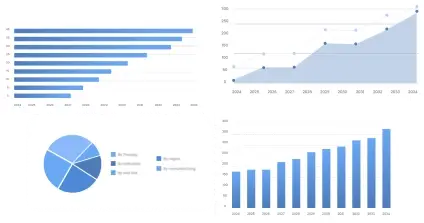
The global in vitro diagnostic (IVD) reagents market size surpassed USD 42.51 billion in 2024 and is predicted to reach around USD 71.93 billion by 2034, registering a CAGR of 5.4% from 2025 to 2034.
| Reports Attributes | Statistics |
| Market Size in 2024 | USD 42.51 Billion |
| Market Size in 2025 | USD 44.81 Billion |
| Market Size in 2031 | USD 61.43 Billion |
| Market Size by 2034 | USD 71.93 Billion |
| CAGR 2025 to 2034 | 5.4% |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
The in vitro diagnostic (IVD) reagents market is experiencing strong growth is due to several trends. An expansion in spending on molecular diagnostics and genetic testing is boosting demand for advanced reagents in precision medicine. The growing use of point-of-care testing reagents, enabling faster and more accessible diagnostics. Artificial intelligence and automation in laboratory setups is improving the speed of testing and also the quality of testing. The market for in vitro diagnostic reagents is also seeing high demand due to testing required during personalized medicine and companion diagnostics, requiring specialized reagents tailored to individual patient profiles. The adoption of liquid biopsy and non-invasive testing methods is expanding reagent requirements.
Two main drivers are the high demand in in vitro diagnostic testing required with a growing global risk of infectious and chronic diseases and the global emphasis on early and accurate disease detection. Despite this, obstacles to growth such as the complicated and expensive manufacturing setups required for advanced diagnostic tests and reagent development, are limiting accessibility in resource-constrained settings. Looking ahead, opportunities include the expansion of diagnostic services in emerging markets with improving healthcare infrastructure and the increasing demand for home-based and self-testing kits, opening new pathways for reagent innovation and commercialization.
| Regions | Shares (%) |
| North America | 40% |
| Asia Pacific | 30% |
| Europe | 20% |
| LAMEA | 10% |
| Segments | Shares (%) |
| Immune Diagnosis | 25% |
| Clinical and Biochemical | 30% |
| Molecular Diagnosis | 20% |
| POCT (Point-of-Care Testing) | 18% |
| Other | 7% |
| Segments | Shares (%) |
| Hospital | 50% |
| Laboratory | 40% |
| Other | 10% |
Published by Vidyesh Swar
| By_Type | 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Immune Diagnosis | 10.63 | 11.07 | 11.52 | 12 | 12.49 | 13 | 13.52 | 14.07 | 14.63 | 15.22 | 15.82 |
| Clinical and Biochemical | 12.75 | 13.35 | 13.98 | 14.63 | 15.32 | 16.04 | 16.79 | 17.57 | 18.39 | 19.24 | 20.14 |
| Molecular Diagnosis | 8.50 | 9.14 | 9.82 | 10.55 | 11.33 | 12.17 | 13.06 | 14.01 | 15.02 | 16.11 | 17.26 |
| POCT | 7.65 | 8.20 | 8.78 | 9.41 | 10.07 | 10.78 | 11.54 | 12.35 | 13.21 | 14.13 | 15.11 |
| Other | 2.98 | 3.05 | 3.12 | 3.19 | 3.25 | 3.32 | 3.38 | 3.44 | 3.50 | 3.55 | 3.60 |
| By_Application | 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Hospital | 21.26 | 22.63 | 24.09 | 25.63 | 27.28 | 29.03 | 30.89 | 32.87 | 34.96 | 37.19 | 39.56 |
| Laboratory | 17 | 17.79 | 18.61 | 19.46 | 20.36 | 21.29 | 22.26 | 23.28 | 24.35 | 25.46 | 26.61 |
| Other | 4.25 | 4.39 | 4.53 | 4.68 | 4.83 | 4.98 | 5.13 | 5.28 | 5.44 | 5.60 | 5.75 |
| By_Region | 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| North America | 17 | 17.83 | 18.70 | 19.61 | 20.57 | 21.57 | 22.61 | 23.71 | 24.86 | 26.07 | 27.33 |
| Asia Pacific | 12.75 | 13.71 | 14.73 | 15.83 | 17 | 18.25 | 19.58 | 21.01 | 22.53 | 24.16 | 25.89 |
| Europe | 8.50 | 8.87 | 9.26 | 9.66 | 10.07 | 10.51 | 10.96 | 11.43 | 11.91 | 12.42 | 12.95 |
| LAMEA | 4.25 | 4.39 | 4.53 | 4.68 | 4.83 | 4.98 | 5.13 | 5.28 | 5.44 | 5.60 | 5.75 |
Last Updated: 26 Aug 2025
Source: Statifacts
| Subsegment | 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Immune Diagnosis | 10.63 | 11.07 | 11.52 | 12 | 12.49 | 13 | 13.52 | 14.07 | 14.63 | 15.22 | 15.82 |
| Clinical and Biochemical | 12.75 | 13.35 | 13.98 | 14.63 | 15.32 | 16.04 | 16.79 | 17.57 | 18.39 | 19.24 | 20.14 |
| Molecular Diagnosis | 8.50 | 9.14 | 9.82 | 10.55 | 11.33 | 12.17 | 13.06 | 14.01 | 15.02 | 16.11 | 17.26 |
| POCT | 7.65 | 8.20 | 8.78 | 9.41 | 10.07 | 10.78 | 11.54 | 12.35 | 13.21 | 14.13 | 15.11 |
| Other | 2.98 | 3.05 | 3.12 | 3.19 | 3.25 | 3.32 | 3.38 | 3.44 | 3.50 | 3.55 | 3.60 |
| Subsegment | 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Hospital | 21.26 | 22.63 | 24.09 | 25.63 | 27.28 | 29.03 | 30.89 | 32.87 | 34.96 | 37.19 | 39.56 |
| Laboratory | 17 | 17.79 | 18.61 | 19.46 | 20.36 | 21.29 | 22.26 | 23.28 | 24.35 | 25.46 | 26.61 |
| Other | 4.25 | 4.39 | 4.53 | 4.68 | 4.83 | 4.98 | 5.13 | 5.28 | 5.44 | 5.60 | 5.75 |
| Subsegment | 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| North America | 17 | 17.83 | 18.70 | 19.61 | 20.57 | 21.57 | 22.61 | 23.71 | 24.86 | 26.07 | 27.33 |
| Asia Pacific | 12.75 | 13.71 | 14.73 | 15.83 | 17 | 18.25 | 19.58 | 21.01 | 22.53 | 24.16 | 25.89 |
| Europe | 8.50 | 8.87 | 9.26 | 9.66 | 10.07 | 10.51 | 10.96 | 11.43 | 11.91 | 12.42 | 12.95 |
| LAMEA | 4.25 | 4.39 | 4.53 | 4.68 | 4.83 | 4.98 | 5.13 | 5.28 | 5.44 | 5.60 | 5.75 |
In vitro diagnostic reagents are critical chemicals used in diagnostic assays, such as immunoassays, molecular tests, and point-of-care devices, to detect diseases, biomarkers, and pathogens. They play an essential role across hospitals, laboratories, and home testing in guiding diagnosis and treatment decisions.
Rising global rates of chronic and infectious diseases, alongside increased awareness of preventive and early diagnostics, are boosting demand. Technological advances, from molecular diagnostics to AI-compatible and automation-ready reagents, are further accelerating growth.
Immunoassay reagents currently dominate due to their widespread application in detecting proteins, hormones, and infections. Molecular diagnostics reagentsused in precision medicine and genetic testing, are the fastest-growing segment. Geographically, North America leads thanks to advanced healthcare infrastructure, while Asia-Pacific is the fastest-growing region, spurred by expanding healthcare access and diagnostics investments.
The rise of decentralized healthcare is fueling demand for point-of-care and home-based testing reagents. Innovations like multiplex molecular assays, lyophilized and eco-friendly formulations, as well as AI-enhanced, automation-friendly designs are improving diagnostic speed, accuracy, and accessibility.
The market faces significant challenges from stringent regulatory landscapes and high development costs, which raise barriers for new entrants. Opportunities are strong, especially in emerging economies, personalized medicine applications, and the growing demand for rapid, early disease detection and companion diagnostics.
To get full access to our Market Insights, you need a Professional Account or a Business Suite.

You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.

You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.

Our customers work more efficiently and benefit from